Spoiler alert: the blood bank is one of my favorite departments in the clinical laboratory, and it's not just for the refrigerators filled with free snacks 🧛🏻♀️
As blood bankers we are constantly learning more and acquiring new knowledge; like the discovery of never-before-described antigens, their antibodies, or groundbreaking research that changes how we test and distribute blood products for our patients.
So in the interest of appreciating the vampires lurking downstairs, and escaping the horrors that persist and await us outside, let's learn some fun, new facts about the blood bank.🩸
Sorry, Urinalysis is Down the Hall.
We've talked previously about the exterior of red cells, learned how our blood cells are studded and spiked with hundreds of proteins, sugars and lipids we call 'antigens,' molecules which are essential for cell recognition, intracellular communication and maintaining physiological function.
One of these antigens, known as Sd(a) or 'Sid' for short, was elucidated and characterized with the help of a volunteer, Sidney Smith, and...the urine of another animal, the humble guinea pig. 
Yes, I really am telling you guinea pig urine has been utilized a non-zero amount of times inside of the blood bank. Stranger words have been spoken, I promise.
While Sd(a) was identified in the late 1960s, the antigen itself was not assigned to a blood group until 2019 when a negative phenotype (absence of the antigen) was finally confirmed!
No Fruit Allowed...Unless..
Laboratories, and the scientists that work within them, are held to strict safety standards and protocols. For example, we do not bring food, snacks, drinks or gum into the lab as these consumables increase the risk for cross-contamination and break aseptic protocol.
Except when fruit is involved, of course.
Enzymes are often employed by blood bankers in clinical investigation to help us solve an especially tricky puzzle, like identifying antibodies that may be reacting in unpredictable ways or ruling-out high-incidence antigens.
Blood bank enzymes are powerful proteolytic tools; super-small chemicals that work fast to 'snip' specific structures on the surface of a cell, like scissors cutting away at a piece of paper or a shell of plastic wrap.
Enzymes work in two different ways, either by destroying certain antigens or enhancing the reactivity of others. We then compare the reactivity of patient plasma with enzyme-treated vs. untreated red cells. This helps blood bankers uncover valuable clues about the antigens that are present or which antibodies may be reacting to them, important information that helps us solve the puzzle and get blood to our patients quickly.
...But where do the enzymes come from?
Fruit! 🥭🍍
- Ficin is derived from figs.
- Papain is derived from papaya.
-
Bromelin is derived from pineapples.
Most blood banks and hematology laboratories today order their enzymes as ready-made reagents from manufacturers. However, there are still a number of reference labs or sentinel facilities that produce their own, in-house, from whole fruit.
You Get What You Get (Terms and Conditions Apply)
We know that our blood type is determined by our inherited genetics; you get some DNA from your paternal parent and you get some more from your maternal parent, and the resulting mish-mash of nucleotides are the ultimate deciding factor for which sprinkles get attached to the icing on your personal oxygen donuts.
Generally, your blood type will remain consistent and reliable throughout your lifetime. If you type AB, Rh+ as a 2 year old you will very likely be an AB, Rh+ 22 year old a couple of decades down the line.
...But you may have noticed I said generally, alluding to the possibility that this isn't always the case.
Maybe you remember a past case we discussed, how the proteins expressed by gastrointestinal bacteria in our microbiome can confound in vitro testing, making a patient's blood type appear to have changed unexpectedly. While the Acquired B phenomenon can result in a blood type appearing to change, this is an in vitro phenomenon and does not represent a true hematological event.
On the other hand, a bone marrow transplant can result in a true change of a phenotype! This is the only known mechanism by which a patient's blood type can undergo conversion.
When a patient undergoes a bone marrow transplant, their own marrow is 'wiped out,' leaving a fresh, clean slate for the new marrow to engraft and get to work churning out new cells for the body. BM transplants may be used for a number of hematologic conditions, but treatment for bloodborne cancers (such as leukemia or lymphoma) are the most common.
Unlike transfused blood or implanted organs, marrow transplants are not matched for patient blood type because we have other markers at our disposal to match donors to recipients. This means a patient's best match may have a different blood type than they do.
Not to fear.
When the hematopoietic stem cells, immature precusors, that comprise bone marrow are transplanted into the inner hollow of the bone, they get to work utilizing the DNA from the BM donor to make new blood cells. The patient's bone marrow was also previously depleted, getting rid of all white blood cells that they'd be producing to attack 'foreign' red cell antigens.
These two factors together mean that the body will convert to a new blood type, while failing to mount an immune response to the antigens that would be previously recognized as foreign and destroyed.
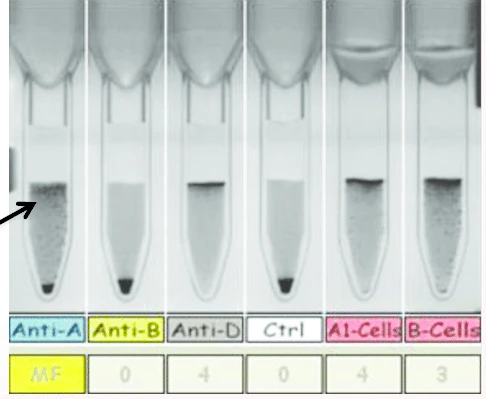
This conversion process takes time. The old blood cells are cleared out over 120 days while new ones are produced, so patients will be left chimeric- holding two separate and distinct populations of red cells within their circulation at the same time. 🤯
Here's an example of what this could look like in gel-phase testing. Note the mixed field reaction for Anti-A, indicating that some cells have the A antigen while others do not.
Once Again, the Answer is Microbiology
Rest assured, I am not in the least bit biased when I say that everything always comes back to bacteria eventually. The weather outside, the snacks you crave late at night, even the ways we react to common allergens are all determined (at least in part) by microscopic organisms that we need powerful lenses and noxious, aromatic stains to see.
So maybe you won't be too surprised when I tell you that a lot of the antigens on our red cells have been found to originate in...yeah. Microbes.
The antibodies that react to the A antigen are cross-reactive with multiple epitopes of the influenza virus. This almost makes some intuitive sense, we know influenza has virulence factors that attach to and agglutinate our red blood cells (referring to hemagglutinin, the H antigen).
On the other hand, antibodies that recognize the surface proteins of E. coli bacteria are cross-reactive with Type B red cells. We briefly touched on this when we discussed Acquired B in a previous post.
Other antigens are implicated too- pollens or fructans from plants, other viral antigens such as capsid proteins, not to mention sugars that we've found in soil or other environmental samples.
Regardless, my favorite has to be the bacteria. Don't worry, I promise I'm not biased.
Mosquitoes and Malaria
Bacteria aren't the only organisms that have played an important role in driving the evolution of the mammalian red blood cell.
It turns out another mischievous microbe, transmitted by the tiny bite of a mosquito, is responsible for most of the antigenic variation on our red cells today.
Malaria refers to a collection of diseases caused by species of the parasitic protozoan Plasmodium. These tiny pathogens are incredibly adept at living inside of blood cells, hiding from the immune system and cause devastating infections that result in anemia, bone abnormalities and even death.

So it may fail to surprise you that as malaria has evolved alongside us, so have humans evolved under the selective pressure exerted by the pathogen.
People with Sickle Cell Trait, who bear one copy of the SCA gene instead of two, are resistant to Plasmodium falciparum- the subspecies of malaria that carries the highest mortality rate. This is due to cellular differences in oxygen tension and cell membrane permeability.
Importantly, and contrary to popular myth, patients with Sickle Cell Anemia are not resistant to P. falciparum and face a much greater risk of death.
People with the rare blood type Duffy null, or Fy(a-b-) are resistant to two types of malaria; P. vivax and P. knowlesi. This is because the Duffy receptors are used by the parasite in cell invasion + infection! An absence of these receptors means that these parasites cannot enter the red cell at all.
Less than 3% of all Fy(a-b-) individuals are ethnically 'white', with more than 97% of carriers having African heritage. I've never been one to submit to statistics, which is why I'm always happy to share I am a (very 'white') Duffy null blood banker.
Other red cell abnormalities or groupings, such as B-thalassemia, Dantu blood group members or rare glycophorin mutations, also affect host vulnerability to infections with specific Plasmodium spp.
1 in 6 humans (more than a billion different people) are carriers of red cell abnormalities or mutations in genes for red cell antigens. This startling prevalence means they are among the most common inherited disorders that affect modern humans!
Today, we know that this is due to negative frequency-dependent selection. That's quite the mouthful!
Basically, cells that express low-incidence variants of antigens are both more efficiently recognized and readily distinguished by cells in the host's immune system. This is important for pathogen recognition and immune priming, so antigenic variation was selected for as humans were exposed to disease.
Humans who have rare blood phenotypes are, at a molecular level, better suited to readily detect pathogens and distinguish them from host cells accordingly.
This explains why rare phenotypes, such as Duffy null or Lewis non-secretors, are more likely to carry innate resistance to infectious disease!
So microbiology, whether we are referring to bacteria, viruses or intracellular parasites, has a whole lot more to do with those funky, sprinkle-covered donuts than you've likely been led to believe.